Bottlenecks and Breakthroughs: Lessons Learned from New Vaccine Introductions in Low-Resource Countries, 2008 to 2013
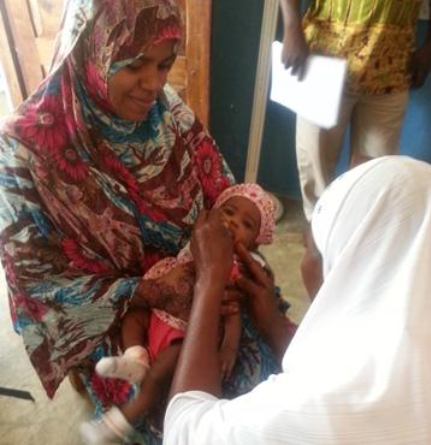
"Popular demand for and acceptance of these new vaccines (primarily pentavalent, pneumococcal conjugate, and rotavirus vaccines) was relatively high due to knowledge and fear of the target diseases for the new vaccines and to successful communication activities."
From 2008 to 2013, the Maternal and Child Health Integrated Program (MCHIP), which was funded by the United States Agency for International Development (USAID), provided technical assistance on the operational aspects of the introduction of 15 new vaccines in 10 Global Alliance for Vaccines and Immunization (GAVI)-eligible countries. The lessons learned from these experiences are shared in this document with the hope that, in the future, they will be useful in supporting smooth and successful introductions of routine immunisation (RI), such as by responding to the call by the global polio eradication initiative to add one dose of inactivated polio vaccine (IPV) to their existing schedule of oral polio vaccine (OPV).
Experiences and practical implications for action from MCHIP's work in the following countries are presented in this paper: Democratic Republic of Congo (DRC), India, Kenya, Malawi, Rwanda, Senegal, Tanzania, Timor-Leste, Uganda, and Zimbabwe. The document covers multiple aspects of new vaccine introduction; however, this summary focuses on communication-related findings, which are highlighted in the following excerpt from the report:
"Demand for new vaccines. MCHIP's experience suggests that most introductions of new vaccines, and particularly PCV [pneumococcal conjugate vaccine], achieved high population awareness and acceptance and, consequently, substantial demand....The ensuing vaccine shortages essentially thwarted the high demand for immunization that had been stimulated by PCV introduction..." In one country, popular knowledge of the availability of a new vaccine against pneumonia created such a spike in demand that there were widespread vaccine shortages that in turn thwarted the high demand for immunisation.
"Communication strategies. High demand also resulted from the extensive and comprehensive communication campaigns conducted in many countries. Key information was distributed through multiple channels: distribution of leaflets, posters, and other print materials; broadcasting of radio and TV messages; and high visibility vaccine launches, media seminars, and stakeholder advocacy meetings."
"National launches were well-publicized, high-profile events that in several instances were attended by the country's president or first lady. Because they attract considerable media attention, launches provide an excellent opportunity to educate the public about the new vaccine and the disease that it prevents and to raise awareness about its availability in the public sector....Local launches were viewed as critical to successful uptake of the new vaccine because they were conducted in local languages, covered by local media, and involved local political and community leaders. In some countries, these local leaders can facilitate or block the local population's acceptance of a new intervention."
"Involving the media. Several countries held seminars or press briefings for the media to enlist their help in informing and providing accurate information to the public. These activities encouraged the media to broadcast TV and radio spots promoting the vaccine, and preempted possible rumors and misinformation about the vaccine's safety. Media representatives often received information packets during the events and were encouraged to follow up by providing coverage of the vaccine introduction. For Tanzania's simultaneous introduction of PCV and RV [rotavirus vaccine] - a particularly high visibility event that drew international attention as well as support from GAVI - a national media seminar was attended by 60 members of the press and other stakeholders and was then followed by zonal-level seminars. These events led to articles being published in local newspapers in various provinces and cities, interviews with Regional Medical Officers on local radio stations, and the airing of promotional spots on local radio and TV stations."
"Strategic engagement of stakeholders. To increase awareness and acceptance of the new vaccine, several countries convened advocacy meetings with stakeholders....In Kenya, such meetings were held for PCV-10 introduction at the national, district, and sub district levels, with participants including representatives from the MOH [Ministry of Health], other government offices, nongovernmental organizations (NGOs), and the community..."
"Developing a basis for key messages and content. Some MCHIP-assisted countries conducted special studies to gain an understanding of common beliefs and attitudes toward the target disease, the new vaccine, immunization, barriers to vaccine uptake, and effective channels of communications. In Kenya, a nationwide knowledge, attitude, and practice (KAP) household survey was conducted both with households and with health workers, community leaders, and journalists....A different approach was used in Rwanda: a small-scale rapid assessment was conducted and analyzed in two weeks. The assessment consisted of focus group discussions with a relatively small number of mothers and health workers....Information gleaned from socio-behavioral studies has been used to inform IEC [information, education, and communication] materials and health worker training to effectively address parents' concerns - for example, about side effects and how to respond to them as well as concerns about their children getting two injections at the same time (e.g., PCV and pentavalent vaccine)."
"Challenges with communication campaigns. In several countries, PIEs [post-introduction evaluations] and monitoring visits found that immunization programs were unable to print and distribute sufficient IEC materials, including materials translated into the appropriate local language, in time for the vaccine introduction. Because of inadequate funding, some IEC materials were still not printed two months after the vaccine introduction in one country and 11 months after the introduction in another".
"Another problem was that high-level government officials and political leaders speaking at public events sometimes gave out incorrect information. In two countries, speakers at national events said that all children under five years of age should receive PCV. These remarks were picked up by the mass media and may have contributed to out-of-age children receiving the vaccine. This experience points out the wide reach and power of well-known figures to create demand for immunization, but it also underscores the need for speeches to be written or reviewed by the immunization program or technical partners to ensure that they are accurate and 'on message'."
Amongst the lessons learned:
- "Immunization programs should anticipate a surge in demand generated by communication activities around the time of a new vaccine introduction....They should prepare by stocking extra supplies of the new vaccine (if possible), and in areas with large numbers of unvaccinated children, they should increase supplies of other infant vaccines as well. This will enable the program to 'catch up' these children and will improve overall immunization coverage rates.
- Conducting special activities to engage the media and elicit their support can be critical, both to create demand for the new vaccine and to preempt potential crises by dispelling rumors or misinformation spread by less responsible elements of the popular press.
- Efforts ranging from nationwide KAP surveys to small, rapid qualitative assessments can be invaluable for developing effective, relevant communication messages that are responsive to people's real needs and concerns. It is critical that these assessments capture the views not just of parents but also of frontline health workers.
- Engaging national and local-level political leaders in vaccine launches and media seminars can be an effective way to generate demand for a new vaccine. However, their messages should be vetted with immunization or public health officials to ensure accuracy and consistency.
- The establishment of an independent committee of well-respected experts to guide the immunization program in introducing a new vaccine can help reassure the public and the media about the vaccine's safety and ensure acceptance of the vaccine."
Email from Mike Favin to The Communication Initiative on February 23 2015; and MCHIP website, accessed March 4 2015.
- Log in to post comments












































