Lessons Learnt during the National Introduction of Human Papillomavirus (HPV) Vaccination Programmes in 6 African Countries: Stakeholders' Perspectives
African Institute for Development Policy, or AFIDEP (Abdullahi); University of Cape Town (Abdullahi, Hussey, Wiysonge, Kagina)
Slow uptake of new vaccines in Africa is well documented; the human papillomavirus (HPV) vaccine is no exception. Despite the HPV vaccine's proven safety, efficacy, and cost-effectiveness, only 8 African countries had reported HPV vaccine introduction at a national level by the end of 2016. By purposively selecting key stakeholders involved in the roll-out of the HPV immunisation programme from 6 of these participating countries, this study sought to investigate experiences, challenges, and lessons learned, with the hope of revealing context-specific knowledge that could be useful to other African countries planning to nationally introduce HPV vaccines.
The researchers conducted a cross-sectional study between 2015 and 2016. From the 8 African countries that had implemented a national HPV vaccination programme (Libya, Lesotho, Rwanda, Uganda, South Africa (SA), Botswana, Mauritius, and Seychelles), one key representative (convenient sampling) was invited to participate. Key stakeholders were defined as members of the National Immunization Technical Advisory Group (NITAG), senior officials from the departments of health at national level, and representatives from international organisations, such as the World Health Organization (WHO) and United Nations Children's Fund (UNICEF). Mixed methods (quantitative and qualitative) were used for data collection and reporting. The researchers obtained results from 6 of the 8 targeted countries.
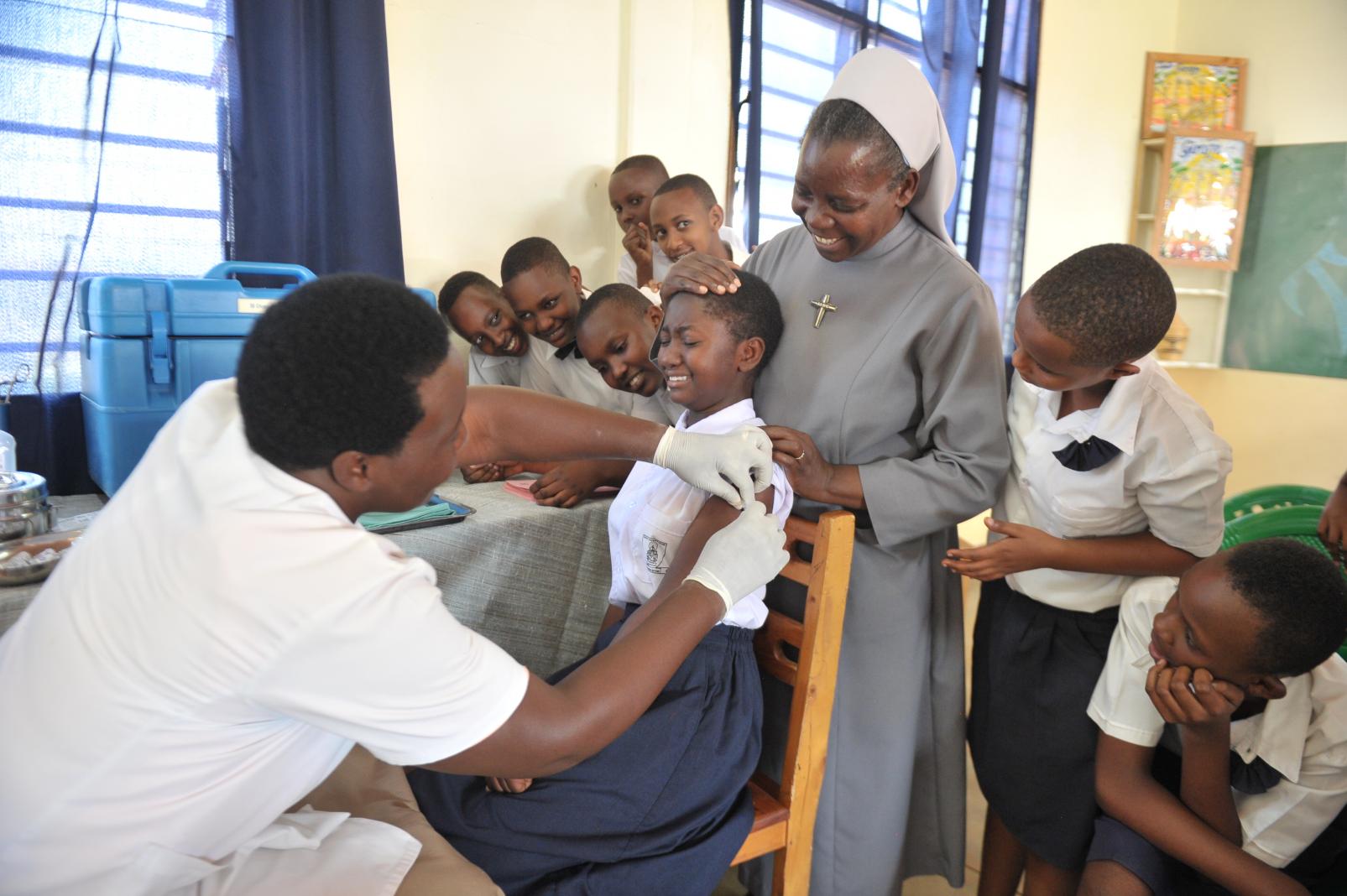
The challenges reported during HPV vaccination programmes, which were similar in all 6 countries, were: identifying the intended population, using a school-based vaccine-delivery strategy, obtaining political support, integrating HPV vaccination with existing school health programmes, and engaging multiple stakeholders. For example, unlike an established Expanded Programme on Immunisation (EPI), where delivery of vaccination services is routine and generally accepted, HPV vaccination is different in many ways, among these being the intended population (adolescents) and delivery through schools. Thus, engagement of multiple stakeholders - parliamentarians, key officials from the departments of education, school heads and teachers, learners and parents - was required, and it was reported to be operationally demanding and time consuming.
Lessons learned included:
- In school, identifying eligible girls for HPV vaccination based on grade/class is more feasible than by age. The proportion of the missed population needs to be quantified.
- School-based delivery strategies can be successfully used to deliver HPV vaccines to learners in African countries with high primary school enrolment. In case of missed vaccination opportunities, healthcare facilities can be used as venues for catch-up vaccinations.
- Obtaining political support during the implementation of the HPV vaccination programme is crucial. Political endorsement by national and district government leaders is also critical for community acceptance.
- Integrating an HPV vaccination programme with existing school-based health programmes is a logistical challenge, but also an opportunity (e.g., to piggyback on other interventions, such as vision screening).
- Adequate preparation for engagment of diverse and relevant stakeholders, including teachers and parents, is crucial.
- Additional support from local and international partners is needed to ensure sustainability of HPV vaccination programmes. Support by local partners is crucial to ensure HPV vaccines reach the hard-to-reach populations in a timely manner.
In conclusion, the researchers "propose collaboration among African countries to document and share the lessons learnt during the introduction of HPV vaccination." Collaborations that document and share immunisation research information can be "platforms to foster further discussions on the introduction of other adolescent vaccines."
South African Medical Journal 2020;110(6):525-531. DOI:10.7196/SAMJ.2020.v110i6.14332. Image credit: © UNICEF/UN0261438/Rusanganwa
- Log in to post comments












































